Welcome back to my blog! I can’t believe I only have one more week left at my incredible internship at the Dorris Neuroscience Center in San Diego. I have had such an enjoyable experience in the lab assisting my mentor Cailynn Wang with her research on tissue clearing and drug distribution. This week I was able to start my own experiment and run through the CATCH pipeline steps all by myself.
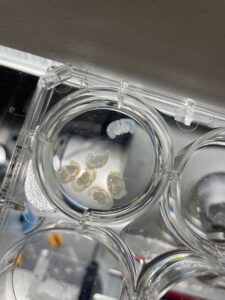
This week was back to normal and the week began with the typical Monday lab meeting. This week’s edition was a journal club, which is where a few lab members present a recently published paper. The topic for this week was the effects of sleep (REM, non-REM, and awake) on the release of growth hormone in mice. Although much of this paper’s content went over my head, it was very interesting to see how everyone gathered to learn something new and weren’t afraid to ask questions when they didn’t understand something. It proved to me that although they are PhD students and post-docs, they still don’t know everything and there is always room to grow and learn more. After lab lunch, Cailynn and I went down to the vivarium armed with the drug cocktail I had made. It consisted of the drug (I chose pargyline), DMSO, saline, and Tween-80. After weighing the two mice, (each was over 30 grams, which is HUGE for mice!), I injected the first mouse with the vehicle solution through an IP (intraperitaneal) injection in their abdomen. The second mouse was injected with the active drug but struggled quite a bit and even managed to kick the needle out at one point. We then had to wait three hours before I could perfuse the mice in order to allow sufficient time for the drug to travel through the blood and bind with the receptors in the brain and other organs. When the time was up I anesthetized the mice in isofluorine and then pinned them to the tray. I then removed the mice’s ribcages and diaphragms in order to access their hearts and place the needle in the left ventricle. I did the perfusions completely by myself, which was a little scary and daunting at first, but I successfully perfused both mice with PBS followed by PFA. After the perfusions I carefully harvested the mice’s brains, hearts, kidneys, and livers. Although I am not as fast or clean as Cailynn when I perfuse and dissect, I am certainly getting better each time I practice! Finally, I ended the day by placing my newly collected organs into a tube filled with 4% PFA so the tissues could continue to fix and become more solid overnight.

The organs I harvested for my experiment with pargyline! One tube contains the organs from the vehicle mouse and other from the one that received the active drug.
Tuesday was a pretty chill day and I spent my morning running another CLICK reaction for one of Cailynn’s experiments. Then, in the afternoon I changed that sample to a PBST-EDTA buffer and washed it for the usual three times. I then stained that sample with the primary antibody, which binds to the proteins that the drug binds to, before leaving it on the coldroom shaker overnight to incubate. I ended the day by venturing into the vivarium on my own and weighing nine mice for Cailynn’s pre-blocking experiment on Wednesday. Pre-blocking means injecting the real, parent drug into the mice and then later injecting the lab-made drug probe (with an alkyne handle). If the pre-blocking is done correctly and the probe is a true representation of the parent drug, the labelling of the probe will be reduced because the sites where is binds have already bonded to the parent drug.
On Wednesday I returned to my collected organs. Cailynn decided that the best option for sectioning would be with the Cryostat, even though it’s finicky, because the vibratome had been completely booked for the next three weeks. Before tissue can be sectioned with the Cryostat, however, it must first be dehydrated and frozen. This dehydration is achieved by placing the tissue in a sucrose buffer that draws out the water in the tissue through osmosis. Dehydration is a crucial step because when the tissue is frozen, if water molecules are present, ice crystals will form that alter the tissue’s structure and damage its integrity during sectioning. I spent the rest of the morning washing Cailynn’s 2-brain sample that had been incubating in the antibody in PBST. I also continued the process of immunohistochemistry (IHC) by mixing the secondary antibody (a 1:500 dilution with PBST) and then adding the tissue to that secondary antibody and leaving it on the warm shaker to rest overnight. In the afternoon Cailynn was busy with her own pre-blocking experiment and doing injections and perfusions, so I worked on some college applications and essay writing.
On Thursday I arrived at my usual time of 9:30 and Cailynn was not yet in the lab. However, she had told me I could wash her antibody sample and mount it by myself. I grabbed the sample off the shaker and proceeded to wash it three times with PBST, (each time for 15 minutes). I then changed the sample into a well filled with the DAPI stain (which identifies nuclei) and put it on the shaker for another 15 minutes. Unfortunately, the lab was having their pipettes calibrated today, so not a single pipette was anywhere to be found this morning. This meant I had to pour the PBST and DAPI, which, although possible, was a little messy. After the DAPI staining, I perfectly mounted the two slices and labelled them appropriately. The purpose of this mini-experiment was to determine whether the antibody used to locate the receptors was effective and “good”. I came back after lunch to find Cailynn preparing to image my slide. We used the confocal microscope and a light wavelength of 488 micrometers to highlight the antibody (receptors) and a wavelength of 647 micrometers to identify where the drug bonded. Theoretically, (in ideal conditions) the antibody and drug should have identical binding patterns. Unfortunately, there appeared to be lots of non-specific binding with the antibody and Cailynn concluded that it was a “bad” antibody. She has faced many challenges with finding the right antibody and I learned that there are lots of failures in science where experiments don’t yield the desired/expected result. The key is to be resilient and try again! While imaging, I also learned a lot more about white matter bundles in the brain and what they do. White matter is the broad term for the axons of neurons that are responsible for transmitting signals in the brain.
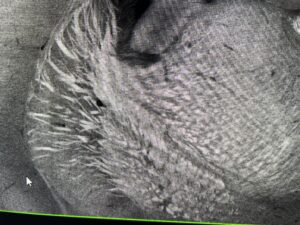
White matter bundles that are like giant highways of myelinated axons that serve to carry neurotransmitter signals across brain regions. They act as the brain’s wiring and communication system
Friday began with a chill morning. I started writing my blog and checked on my sample that was dehydrating in the sucrose solution. The brains had sunk to the bottom, meaning they were sufficiently dehydrated and could now be frozen in OTC and sectioned! Before this could be done, however, I attended a seminar on the mechanobiology of labor and delivery in the lab’s auditorium. It was presented by a post-doc and PI (who had one the Nobel Prize in Physiology/Medicine in 2021!) and was very interesting. After lunch, I froze my brains in the OTC over dry ice and let them rest for 30 minutes. Then, Cailynn and I mounted them onto the Cryostat and I began sectioning. My mentor prefers the vibrato to the Cryostat because the Cryostat is finicky and temperamental, but I started out slowly and learned its tricks. Although it took me longer than the other seasoned lab members, I successfully sectioned both of my brains and the slices were so pretty! I was so proud of myself! Even my mentor said they were very pretty, especially considering they were made using the Cryostat machine. I ended the day on a high note and I can’t wait to finish clearing and imaging my own sample next week! I can’t believe there’s only one week left.
My last Saturday in San Diego was well spent. I went on another run this morning around Mission Bay, (the weather was much cooler today and there was a nice breeze), and then Reign, Shiloh, Azari, Maiya and I spent the afternoon and the peaceful Torrey Pines Beach. We ended the day by dressing up to go to dinner at Chili’s and then taking sunset pictures on the beach. It was an amazing day! I am so excited to take advantage of some more California activities tomorrow and celebrate our last weekend together in San Diego. I can’t wait for next week!


There are no comments published yet.