Welcome back to my blog! For those who aren’t familiar with my internship, I have been working under my incredible mentor Cailynn Wang in the Ye Lab within the Dorris Neuroscience Center. Cailynn’s work focuses on using CATCH (clearing-assisted tissue click chemistry) to map drug distribution in brains in order to create safer and more effective drugs in the future. CATCH allows scientists to determine whether their drug is binding to the correct receptor sites in the brain and see any other unwanted sites it is also binding to that could cause undesirable side effects. Moreover, her research with CATCH and the microscope imaging that accompanies it allows scientists to determine the correct dosage for drugs because they can see how much is binding in the brain.

Cailynn’s lab bench! I have learned that labs are very busy workspaces with lots of things happening all at once.
Week 2 has been just like Week 1: excellent! On Monday I began by attending the weekly lab meeting where one of the grad students, Logan, presented his data update. Later, I helped Cailynn change buffers (put into PBST) for the lung tissue that we had sectioned last week. Changing buffers is a crucial step in the CATCH process because the washing process removes lipids and excess unbound molecules from the tissue that cause impairment of the clearing process and lead to less ideal transparency. PBST (or phosphate-buffer solution w/ Tween20) is used for the first washing cycle and reduces background noise (unbound molecules) which leads to a cleaner and more specific drug signal. In addition, PBST maintains the tissue sample’s integrity so it is less likely to be damaged when mounting or viewing later on. Today I also spent the majority of the afternoon familiarizing myself with all the various forms of histology, which is the study of the microscopic structure of tissues or cells. The primary histology methods that are utilized in the lab are IHC (immunohistochemistry), mass spectrometry, and microscopy. IHC consists of staining the tissue sample with antibodies and fluorescent dyes that label specific proteins/antigens in the tissue. The (primary) antibodies that the samples are incubated with recognize antigens or proteins in the tissues, and the secondary antibodies contain a fluorophore or enzyme complex that binds to the primary antibody and allows scientists to view where the specific protein is.
On Tuesday I arrived early at the lab in order to prepare for the busy day ahead! Cailynn’s experiment that had been delayed last week was on schedule for today. We began by weighing and injecting her cohort of 22 mice, a process that took over two hours because we had to wait for ten minutes in between each mouses’s injection. Today, the experimental drug was sertraline, which is similar to ketamine in that it shows promising antidepressant effects but different because in mainly binds in the dorsal raphe region of the brain, rather than the LHb (lateral habenula) where ketamine binds. Scientists discovered that sertraline primarily targets the dorsal raphe region through the use of IHC, which showed a high presence of SERTs (serotonin transporters). The dorsal raphe is a major source of serotonin and helps control mood and anxiety, meaning its regulation is crucial for treating depressive disorders. In addition, the experiment included four vehicle mice who only received a saline injection in order to establish a control group. The variable in the experiment was the amount of time between injection and perfusion, and whether a PBS perfusion was performed in addition to the PFA (fixative) perfusion. This afternoon I also got the chance to watch “surgery” on the mice brains. In order to control experiments better and reduce the risk of confounding variables, researchers have to ensure that their experimental drug is binding to a specific region of the brain because of the target receptors and not randomly or because of another receptor. In order to test this hypothesis, a virus is injected (through “surgery”) into the mouse’s brain that knocks out the target receptors and allows scientists to statistically determine whether or not their drug is binding primarily due to the target receptors. In surgery, the mice are placed under anesthesia and have their hair removed with NAIR (which works shocking well) before having their skulls cut into and their brain exposed. Then, an extremely precise drill is used to create tiny holes in the mouse’s hypothalamus through which the virus is injected at a stead rate of 100 mL/minute. After surgery, the mice have their skin sewn up and are placed back in their cages and are good to go!
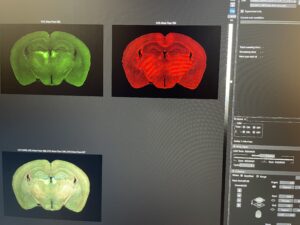
Using a microscope, scientists can see where the drug-bound molecules (bound through CLICK reaction) are binding, (and to what type of cell/protein). If two signals overlap, it means that the drug is binding to that specific type of cell/protein.
On Wednesday, Cailynn and I continued the CATCH process by starting the CLICK reaction. First, we used microliter pipettes to create a main mixture of PBS, DMSO, and an azide reagent. Then, we aliquoted 195 microliters into two separate test tubes with the sample brains and added another prepared mixture of CuSO4 and BTTP. These two mixtures were then left on the shaker for one hour before having their buffers changed to PBS-EDTA three times. DMSO (dimethyl sulfoxide) is used as a solvent for the azide reagent and also enhances solubility and stabilizes the copper (I) ions. CuSO4 (or copper (II) sulfate) is used as a source of copper (I) ions and is used in conjunction with sodium ascorbate to initiate the CLICK reaction. Finally, BTTP is a ligand used to accelerate the reaction and improve the reaction rate, which minimizes copper toxicity in the tissue. After lunch, (and after washing the recently CLICK-ed brains three times), I helped Cailynn use IHC to stain the brain samples with antibodies that will bind to the antigens (specific proteins, in her experiment they are NMAD receptors) in order to identify whether or not the ketamine tissue is actually binding to the NMDA receptors. Using a microscope, scientists can see where the drug-bound molecule (bound through CLICK reaction) are binding, (and to what type of cell/protein). If two signals overlap, it means that the drug is binding to that specific type of cell/protein.
On Thursday, in honor of Juneteenth, the lab was closed and we were all given the day off. To take advantage of this, the other San Diego interns and I spent our day relaxing at Ocean Beach and visiting the local thrift stores. (We also made sure to get our daily consumption of boba and check out a famous bookstore!)
Friday was a pretty chill day at the lab and I spent most of the morning changing washes on our brain samples after their 48 incubation with the antibody stains. This washing removes excess lipids and reduces non-specific bindings. In the afternoon, Cailynn and I prepared the agarose gel and mounted her 22 brain hemisphere samples from Tuesday, then left it to cool before being sectioned by the vibratome. The vibratome is an efficient way to slice multiple samples at a time while maintaining the tissue integrity. It also allows for free time to mount more slides. Cailynn says I am mastering this aspect and entrusted me to mount two brain slices for her and a slide with nine lung samples!
On the weekend the other pinterns and I took the chance to be tourists and visit more of San Diego’s iconic attractions. On Saturday morning we visited the huge farmers market in Little Italy where we indulged in many sweet treats, including a delicious coconut sundae!
Other fun things we have done include a (very off-key) karaoke night, lots of board games, a very competitive ping-pong tournament, and hearing all about the adventures of Bellamy and Rihanna (the cell cultures that Shiloh and Azari created and are literally obsessed with (they wrote them birthday cards 😀 )).
I am having so much fun in San Diego and I cannot wait for what next week holds!


Wow! It sounds like you’re really learning a lot and contributing to an important study. Glad to see that you’re checking out the markets … never went to the one that you did! See you soon.