Hi again! I’m Shiloh and this is week 5 at the Briney Lab at Scripps Research Institute in San Diego, California. Quick Refresher: the Briney Lab operates within the Immunology and Microbiology department at Scripps as a subsection of the Burton Laboratory. Its members work to analyze human immune responses to infectious diseases like HIV, Lassa, Ebola, and SARS-CoV-2 in order to identify and develop antibody proteins capable of effectively targeting these pathogens in a process known as “reverse vaccinology”. Last week we analyzed our antibody production levels and this week we were finally able to begin putting them into action.
We worked with Sean on some of the earlier stages of plasmid preparation as well as with Ben Nemoz: Azari’s mentor who arrived this week. He gave us a full secondary run down on PCR (polymerase chain reactions) while Sean let us work with him on some Gibson Assembly, SDM (site-directed Mutagenesis), PCR, and eventually transfection of the plasmids we produced. A lot of the work Sean does is in silico and his wet lab work reflects is performed to validate the models he builds through the Alphafold pipeline. The 3D models created by Alphafold allows us to examine the exact amino acid chain link that comes into contact with viral trimers during antibody/epitope interactions. The overarching idea is to predict germline mutations that will induct effective antibodies and then guide this process towards the creation of the structures that favor binding. We want to guide the affinity maturation of b-cells so that they begin to naturally create virus targeting antibodies on their own; this way the b-cells can be ‘taught’ to adapt alongside viral mutations and remain effective. Alphafold helps us to develop immunogens that will activate b-cells suitable for this particular evolutionary pathway as well as discover the antibody sequences we want to eventually develop. The coding program can be finnicky though since it’s taught on databases full of currently registered antibodies. This causes it to create a lot of antibody protein structures that fold nicely into a ‘happy protein’ but don’t work effectively as a ‘happy antibody’ should. To lower the effect of this inaccuracy in our studies we can cross reference the sequences produced by antibodies alone versus the sequences created when we view an antibody-antigen complex and essentially “cancel out” the amino acids that make up the chains of the happy proteins and single out the amino acids that are actually changing the protein structure to promote more effective binding. All of this remains purely theoretical in it’s practicality however, until it’s results can be justified through wet lab antibody production and testing.
All in all, the coding and computational work Sean does is extremely confusing; at times I think even he isn’t entirely sure of what he’s doing, but it’s also extremely cool and pushes the boundaries of what’s currently possible in vaccine development. It’s painstaking in it’s current limitations but the AI models are constantly being trained to perform faster, easier, and more accurately. Eventually this in silico form of vaccine design could help to reduce the need for in-vivo (live) experimentation which is costly and occasionally time consuming as well as ethically controversial on occasion.
On that note, in-vivo experimentation was the large majority of what we practiced this week. First, we examined antibody effects on sporozoite motility through the use of confocal microscopy. 311 v 317 with PGT121 as a control. At .1 ug/ul 311 was more effective at reducing motility in the parasites than 317, suggesting it is likely the more effective antibody. However, in earlier clinical mouse studies, 311 and 317 have been shown to have similar potency and breadth levels in neutralizing Malaria. This leads us to believe that the two antibodies may have different mechanisms of action; essentially 311 may work by inhibiting the movement of the parasite where 317 inhibits its ability to invade a cell. Our B6 mice came in on Friday and so we’re now able to analyze our antibody’s capabilities in live specimens otherwise known as “in-vivo”. We have three groups consisting of five mice each for a total of 15 overall. Each and every single one of them has a name despite the multiple warnings we were given from other lab members that naming the mice is a bad idea when it comes to their eventual termination. Group 1 was given doses of the antibody 311 , group 2 given antibody 236, and group 3 left to act as our control after having received an injection of Zika-V1, an antibody that binds exclusively to Zika Virus and as such should have no ability to provide Malaria defense. The antibody dosages are delivered retro-orbitally into the sinus behind the eye which allows for the rapid systemic administration of substances into the bloodstream. Some institutions use tail veins instead but Nate B. prefers to inject retro-orbitally so that’s what we did. The mice are all given anesthesia before hand and sleep through the entirety of their injections. I tagged the mice of each group using ear ‘piercings’ for identification(L, R, LR, LL, none) while Azari prepped and loaded syringes for Nate B. to deliver with. We worked like a little factory line except we were passing around mice and tubes instead of toy cars and cookies. Later on Friday we challenged the mice with sporozoites, which follows the same exact injection procedure but with Plasmodium berghei (a mouse-infecting malaria strain) rather than antibody.
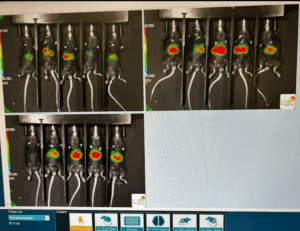
1-311 2-236 3-negative control 2 should have no light to it (the concentrations are still lower scale wise just not as low as we want)
Azari tried to perform one of the retro-orbital administrations on a mouse from group 2 and almost succeeded but couldn’t quite keep the syringe positioned within the sinus throughout delivery. The mouse bled profusely from the eye for a second which freaked us both out since it’s possible to pierce too deep into the membrane there and enter the brain cavity (potentially killing the mouse) but ultimately after a little TLC, the little girl was fine. I thought she did pretty good for a first timer. Because we challenged on Friday, we spent Sunday morning in the lab imaging and analyzing. Azari and I each performed intraperitoneal injections (body cavity) on a cohort of the mice with 100 ul D-luciferin each in order to measure luminescence. The mice are injected with 50 ul on either side of their stomach and then placed in a AMIHTX in vivo imager. The greater the ‘glow’ that’s measured the more infected the mice are but I don’t think we waited long enough between antibody administration and challenging to get truly accurate results. Our 311 antibody should have neutralized the plasmodium yet our imaging showed a live infection. Given that infecting these mice with plasmodium would eventually lead to a prolonged and inhumane death we had to euthanize them after this procedure via cervical dislocation and CO2 exposure. This is all done super humanely, the mice remain under anesthesia for cervical dislocation.
I also had to prepare and practice a part of my internship presentation in front of a small cohort from the labs (Thanks Iszac, Ben, Liendo, and Nate B. for all your constructive feedback!!). Their advice was really helpful and clarifying, so it made me feel a lot better about having to present later this year, even though I was nervous just about presenting for them at first! Also, my final presentation is basically finished now so I don’t have to worry about making the whole thing later on with possibly incorrect information, yay! We also had a nice brunch with Ben, Azari’s second mentor who just got back from France. He’s super nice and also studies quantum mechanics in his free time despite the fact that he’s already a double doctor (MD and PhD). That short talk led to a whole fiasco about multiverse theory based out of Nate B.’s office. It got a bit out of hand and spanned nearly an entire hour since various lab members like Liendo, Charlotte, and Professor Lars Hangartner kept feeding the flames of the conversation. I think Liendo was actually invested in the physics of it all and Lars just wanted to watch us all slowly go insane about the concept of infinity beyond it’s use as a mathematical construct. I’ll die on the hill that the universe is neither linear nor singular though. As always, our weeks end with the splitting of cells.
Outside of the lab we surfed some more (I’m still really bad at it) and played some beach volleyball (not quite as bad at that thankfully). My surfing skills are so terrible in fact, that a complete stranger approached me on the beach after I gave up on my aspirations of becoming a fish and told me that I made her feel better about her own inability to make it past the breakers. At least I provided some free entertainment for Azari, who didn’t even make an attempt. Volleyball was super fun but also super tiring; I love playing court but the sand will suck the life out of you it’s so hard to move. It tired Azari and I out so much we decided to walk two miles to the nearest gas station while our housemates were out so we could get snacks. So worth it: gas station cheese is fire. We’ve also engaged in copious amounts of karaoke, movie watching, beach going, and arts and crafts. This Saturday I slept in late and then our house group headed to Del Mar and “hiked” down a small bluff from the railroad tracks to get to a more secluded part of the beach. We’d visited the more mainstream area before but it was even nicer to hang out on a quieter section where we were able to draw in the sand and hang out by the water relatively undisturbed by tourists and the like. Technically we’re also tourists but after five weeks you really do start to feel like a local! We dressed up in the best we packed and headed out for ‘fancy dinner’ since this will be our last weekend together in San Diego. We ate at Chili’s and made mini canvas paintings there (they all turned out so cute!!), then SPRINTED to the beach to try and catch the sunset for pictures. Don’t judge us too hard, at the end of the day were still a bunch of teenage girls. And our pictures turned out really cute so score! Definitely going on the wall when I get home.
I actually leave for home this Wednesday, so this will be my last full week blog post! As always, thanks for listening in; anyone who made it this far gets good luck for the rest of next week 😉





There are no comments published yet.