Welcome back, everybody. I’m Azari, and I just completed my fifth week at Scripps Research. Just a refresher, I’m working with Nate Beutler, a post-doctoral immunologist who, through antibody discovery and therapeutic design, is working to understand why an effective vaccine for malaria hasn’t been developed yet. This week, my other mentor, Ben Nemoz, returned from his trip to Europe. He’s a post-doc in the United States and holds an MD in France. He splits his time between both places. Ben is a virologist who specializes in HIV research. This week, I got to join Ben in his lab work, repeated some projects from previous weeks, and started on my final project in the lab.
Our confocal microscopy from last week wasn’t as successful as we would’ve liked, so we restarted the process on Monday. We did a serial dilution for each of our antibodies: mAb 311, mAb 317, and mAb pgt121. Antibodies 311 and 317 target malaria sporozoites, while mAb pgt121 targets HIV, making that our negative control. For each antibody, the initial concentration varied but was all at a dilution factor of 10. So our most concentrated samples will immobilize more of the parasite while the least concentrated will immobilize less of the parasite. The whole point of the experiment is to compare the behavior of the malaria antibodies and at what concentration they work best. We found that at .1ul/mg, mAb 311 was more effective than mAb 317. Since the concentration is so small though, they would behave almost identically in vivo. At the end of the day, we split our new adherent cell cultures. I didn’t really have to do any splitting, I just moved the culture to a larger flask.
Tuesday was a bit mundane which is part of the job. Not every day is gonna be full of excitement. We continued and finished our confocal microscopy before going to Costco for lunch with Nate and Yenni. Tuesday was also the day my other mentor, Ben, came back from Europe. He’s a really high-energy and passionate guy so I’m really glad I get to work with him. He’s really humble about the fact that he’s a ‘double doctor’; having a PhD in the U.S and an MD back at home in France. On Wednesday, I got to actually work with him and got to know him a bit better. We did a PCR for a vaccine he’s developing. The vaccine needs mRNA inside an LNP (lipid nanoparticle). In order to get to that step we need to get DNA. To get our DNA of interest, we replicate the complementary sequence then replicate the complementary of that sequence. We end up with an identical sequence to our origin. For PCR we need our enzyme (like the mechanic), our DNA template (the blueprint), nucleotides (TTP; the building blocks), water, 5’ forward primer, 3’ reverse primer, and magnesium. We start off by ‘unzipping’, or denaturing, our DNA sequence at 98°C. The bacteria we’re using for this are native to Yellowstone, meaning they can undergo and withstand intense heat. The nucleotides are bonded with a hydrogen bond. It isn’t very strong so the sequence can easily be broken up at that temperature. We then bind our primers to the start of our complementary 5’ and 3′ at 50°C-60°C. The sequence is annealed to the complementary and the cycle is repeated about 30-40 times for amplification. Growth is exponential and at about the 30-40 range, it crosses the threshold for detection. After finishing amplifying our DNA, we cleaned our final DNA and ran it through a gel. This one is different in that it’s almost completely computational. The gel is premade and we analyze it on a computer. The only manual work is placing the marker and the DNA. Even though we messed up a bit, our DNA turned out basically perfect! Shiloh and I ended the day by splitting both of our cell cultures again. I definitely got the hang of it now. Nate invited us to go surfing with him after work again which was super fun. This time I chose to stay at the beach while listening to music and reading. I did enjoy watching Shiloh fall off her board very frequently!! A girl at the beach approached Shiloh saying that watching her fall made her feel much better about not being very good either.
Thursday was just as laid back as Tuesday. Ben was nice enough to buy us breakfast in the morning and we continued getting to know him. We learned that he likes to study quantum mechanics in his free time which is insane to me because he’s already a double doctor. He also told us a story about him giving a man CPR at 7 am on his way to a big presentation while he was in France. He not only saved a man’s life but he then went on to give an amazing presentation. He recommended a book to me so I bought it and look forward to reading it! The conversation with Ben about quantum physics stuck with me so we continued the conversation with a few other people in the lab. It ended up being an hour-long discussion on the possibility of the multiverse and the concept of infinity. Later Shiloh gave a short presentation to a few people in the lab. There wasn’t any lab for me to do since both of my mentors were working on research papers so I spent the rest of the day on some personal work. Nate and his girlfriend Marie invited us to play volleyball with them at the beach after work so of course we went. I feel lucky knowing a local with insider knowledge of the best spots in San Diego. I feel like a real local now. When we got home Shiloh and I decided to walk two miles to get snacks at a gas station and it was totally worth it. The sunset was absolutely beautiful and the snacks were just as good.
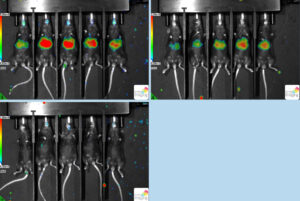
Friday was one of the busiest and truly engaging days I’ve experienced here so far. We began the day by doing minipreps and a Gibson Assembly for Sean. While waiting for Nate to return from a meeting, Shiloh and I successfully split our adherent and suspended cells completely on our own again. Shiloh is a bit worried hers are unhealthy since they were severely overgrown when we checked up on them. When Nate arrived, we performed a challenge on mice. We performed a retro-orbital delivery of Plasmodium berghei into mice to test how well our antibodies protect the rodent. We had 15 mice; 5 for mAb 311, 5 for mAb 236, and 5 for mAb Zikv1. 311 and 236 target different parts of the circumsporozoite protein (protein found on the surface of the malaria sporozoite). Zikv1 is used for Zika virus so that was our negative control. My job was to prep the syringes. The measurements have to be super accurate. First, I pulled the plunger in and out so it’s not as stiff when injecting. I then extracted 100ul of each antibody. It’s important to get rid of all air bubbles. After the bubbles were out I pulled back slightly before carefully pushing the syringe up until a tiny amount of the liquid came out of the needle. Nate injected each prepped antibody syringe into the corresponding mouse. Two hours later we came back to inject the mice with Plasmodium berghei. The procedure was the same but at this point it’s really really important to be accurate and precise. I don’t know why but Nate let me inject a mouse. I injected a mouse from the 236 group. I definitely didn’t do great but it’s fine because it was my first time ever performing a retro-orbital delivery. On one side of the mouse, I pulled out slightly so the liquid leaked out of the mouse. On the other side, I think I poked the wrong place because it started bleeding. Luckily we have four perfectly done mice!! Initially, I was nervous about working with animals but I’ve kinda gotten the hang of it and I understand the purpose of it all. It was so interesting that we finished at around 6 pm and we didn’t even realize. Nate asked if we wanted to come in on Sunday to finish challenging our mice and obviously we agreed. Sunday morning we did a intraperitoneal injection of D-luciferin on the mice to analyze the protectiveness of each antibody. The more the mouse glows, the less protected it was. We found that group 311 wasn’t as protected as we would’ve liked; it’s likely that we were given the wild type of our Plasmodium instead of the transgenic Plasmodium. Since the mice were infected with Plasmodium berghei, we humanly euthanized them via cervical dislocation and CO2 exposure to avoid an unnecessarily prolonged death. Overall the experience was really cool and it’s definitely influenced my decision to continue on a STEM related field.
This weekend is our last weekend in San Diego so we made sure to make the most out of it. We spent all morning at a hidden beach in Del Mar. Thanks Reign for letting us bury you in the sand! We rushed home to get ready for our super fancy dinner at… Chili’s. While waiting for our food we had a quick painting session. After struggling profusely trying to work the card reader we ran over to another beach to take some sunset pictures. We went to my favorite beach, Scripps Beach. It’s the same beach that Nate takes us surfing all the time so it was nice having all the other girls have a fun experience there too. We got a bunch of photos that I will most certainly be putting on my wall when I return home. I’m really coming to terms with the fact that I have to go back home this time next week which is so bittersweet. This summer has been one of the best summers I’ve experienced so far and I get a bit sad at the thought of having to leave it in just seven days. I’m very grateful that I got to not only learn about the ins and outs of immunology but I got to do it all with the most amazing people. Go team SD!!



There are no comments published yet.